The long-term, durable clinical benefit of Opdivo plus Yervoy with two cycles of chemotherapy was observed at three years across patient populations that typically have a poor prognosis, including patients with PD-L1 expression With a minimum follow-up of three years (36.1 months), the dual immunotherapy-based combination continued to show sustained improvement in overall survival (OS), the trial’s primary endpoint, with 27% of patients treated with Opdivo plus Yervoy with two cycles of chemotherapy alive compared to 19% of patients treated with chemotherapy alone at three years (Hazard Ratio 0.74 95% Confidence Interval : 0.62 to 0.87). Late-breaking data to be presented during the 2022 American Society of Clinical Oncology Annual Meetingīristol Myers Squibb (NYSE: BMY) today announced three-year follow-up results from the Phase 3 CheckMate -9LA trial demonstrating durable survival benefits with Opdivo (nivolumab) plus Yervoy (ipilimumab) with two cycles of chemotherapy compared to four cycles of chemotherapy in previously untreated patients with metastatic non-small cell lung cancer (mNSCLC). Claim your 1-week free trial here.ĭual immunotherapy-based combination resulted in clinical benefits across key subgroups of mNSCLC patients with high unmet need, including those with PD-L1 expression Importantly, we saw long-term benefit among patients with high unmet needs, such as those with PD-L1 expression <1% who tend to have worse outcomes and fewer treatment options.Get inside Wall Street with StreetInsider Premium. “The three-year data from CheckMate -9LA demonstrate the ongoing durability with early disease control following treatment with the combination of nivolumab plus ipilimumab, with a short course of chemotherapy. Paz-Ares, M.D., Ph.D., CheckMate -9LA study investigator and chair of the medical oncology department, Hospital Universitario 12 De Octubre in Madrid, Spain. “While immunotherapy treatments have greatly improved outcomes for people with metastatic NSCLC, many patients, particularly those with low PD-L1 expression, are unfortunately not achieving durable, long-term survival,” said Luis G. These data will be featured in a late-breaking poster presentation (Abstract #LBA9026) at the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting on June 6, 2022, from 9:00 a.m. No new safety signals were observed with Opdivo plus Yervoy with two cycles of chemotherapy with extended follow-up in the CheckMate -9LA trial. In an exploratory analysis, a positive trend for OS benefit was also observed with Opdivo plus Yervoy with chemotherapy among patients with certain tumor mutations, such as STK11. Squamous histology: Among those with squamous histology, the OS rate was 24% for patients who received Opdivo plus Yervoy with chemotherapy, compared to 11% for those who received chemotherapy alone. PD-L1 less than 1%: Among patients with tumor PD-L1 expression less than 1%, the OS rate was 25% for those treated with the dual immunotherapy-based combination vs. 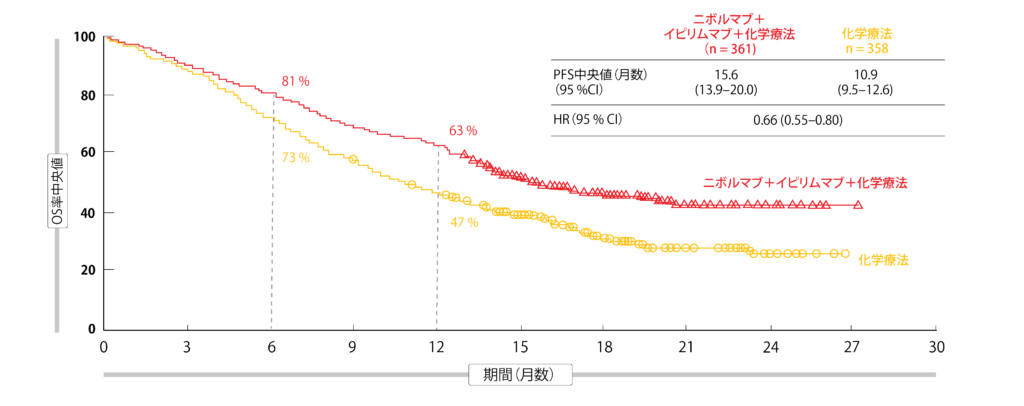
The long-term, durable clinical benefit of Opdivo plus Yervoy with two cycles of chemotherapy was observed at three years across patient populations that typically have a poor prognosis, including patients with PD-L1 expression less than 1% and squamous histology.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
